Answer Summary
DME providers operationalize 2026 CMS rules by embedding compliance requirements directly into repeatable workflows—moving beyond static policies to ensure documentation and authorization integrity are validated in real-time. This shift is mandatory as CMS increases its use of automated data analytics for targeted audits, with industry leaders targeting a 20% reduction in audit-related recoupments through continuous readiness. By treating compliance as operational infrastructure rather than a periodic obligation, providers protect their cash flow from the financial volatility of reactive regulatory responses.
Introduction: Why “Knowing the Rules” Is No Longer Enough
For many DME providers, compliance has traditionally been treated as a regulatory requirement rather than an operational discipline. Leadership ensured policies existed, staff completed annual training, and audits were addressed when they occurred. This approach was imperfect, but it was often sufficient in an environment where enforcement was episodic and payer behavior relatively predictable.
In 2026, that model no longer works.
CMS compliance expectations now extend far beyond written policies. Enforcement increasingly focuses on how rules are executed within daily workflows, not whether providers can cite regulations. Documentation integrity, authorization discipline, supplier standards, and audit readiness are evaluated continuously, often through automated review rather than isolated audits.
DME providers must therefore shift their mindset. Compliance is no longer about awareness—it is about operationalization. The organizations that struggle in 2026 are not those unaware of CMS rules, but those that have failed to embed those rules into repeatable, controlled processes.
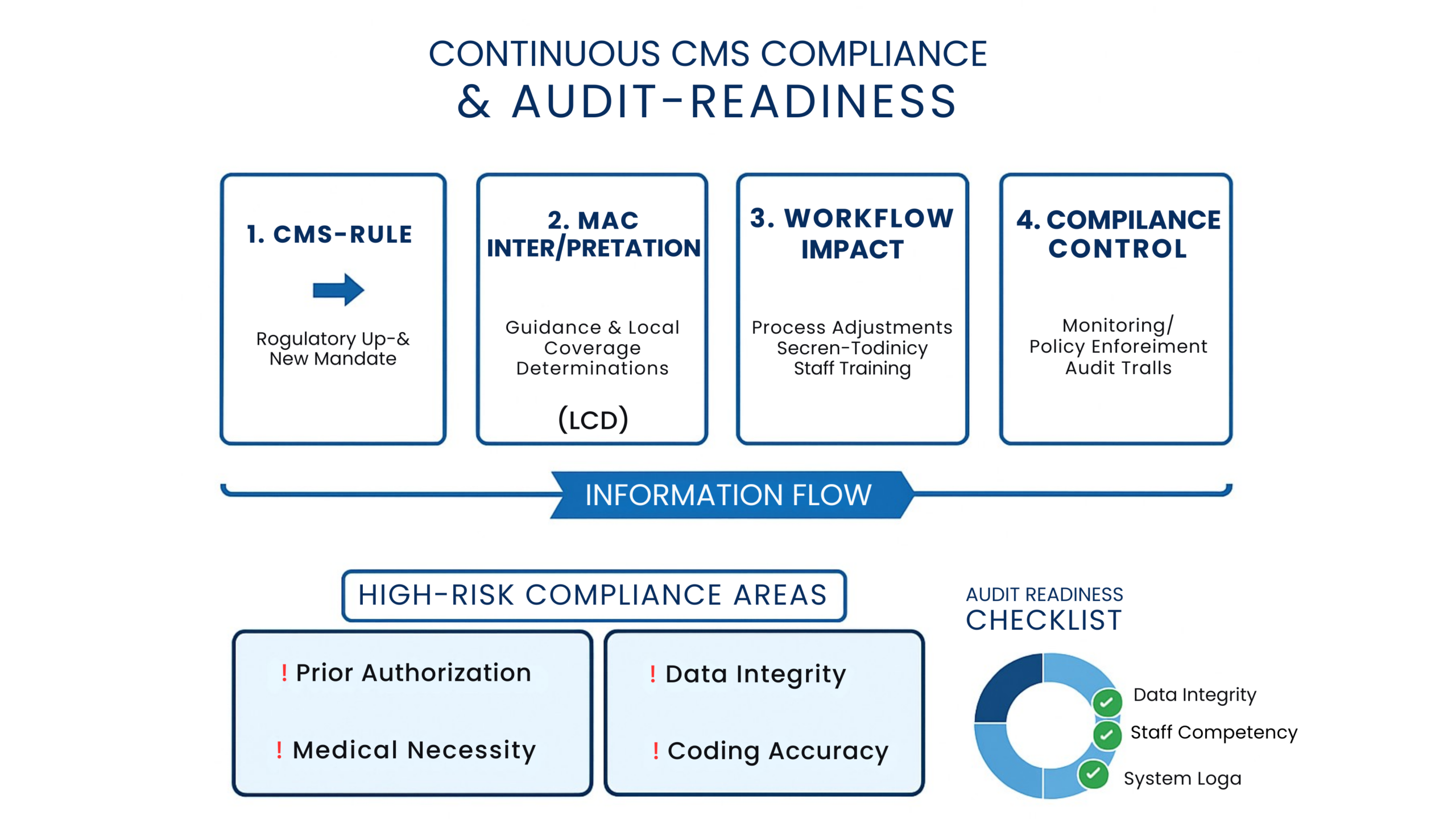
How CMS Rule Changes Actually Reach DME Operations
One of the most misunderstood aspects of CMS compliance is how regulatory changes translate into day-to-day operational impact.
CMS publishes rules and guidance at a federal level, but enforcement rarely occurs directly from CMS to the supplier. Instead, rules flow through multiple layers:
- CMS publishes regulations and interpretive guidance
- Medicare Administrative Contractors (MACs) interpret and operationalize those rules
- Payers align policies and review logic accordingly
- Providers experience the impact through denials, audits, and recoupments
Breakdowns occur when providers focus only on the initial CMS publication without tracking how those rules are interpreted downstream. A rule that appears benign at a high level may result in stricter documentation requirements, narrower authorization parameters, or expanded review criteria once implemented by MACs and payers.
Compliance failures often stem not from defiance, but from lag—a gap between regulatory change and operational adjustment.
Documentation Standards as an Enforcement Tool
Documentation has always been a compliance requirement, but in 2026 it has become a primary enforcement mechanism.
CMS and its contractors increasingly evaluate documentation not only for completeness, but for internal consistency and sufficiency. A physician order may exist, but if supporting documentation does not clearly justify medical necessity for the specific item billed, the claim is vulnerable.
Under 42 CFR § 424.57, DMEPOS suppliers are required to maintain records that substantiate the medical necessity and delivery of items billed. In practice, this means:
- Documentation must align across intake, delivery, and billing
- Medical necessity narratives must support the specific equipment provided
- Records must be readily retrievable and defensible during review
Providers that rely on minimal documentation standards often find themselves repeatedly correcting the same issues after denials or audits. In contrast, organizations that embed documentation sufficiency checks into workflows reduce exposure significantly.
Supplier Standards and Operational Accountability
CMS supplier standards are frequently cited but inconsistently applied. Many providers view them as static requirements rather than operational controls.
In 2026, enforcement trends indicate increased focus on how supplier standards are executed in practice. Common areas of exposure include:
- Intake processes that allow incomplete records to advance
- Delivery documentation that lacks clarity or consistency
- Record retention practices that rely on individual memory rather than system controls
The phrase “we’ve always done it this way” often signals risk. Supplier standards are not suggestions—they define operational accountability. Providers that translate these standards into concrete workflow requirements are better positioned to withstand scrutiny.
Audit Readiness Is No Longer a Point-in-Time Event
Historically, many DME providers treated audit readiness as a periodic effort. Files were organized when an audit was announced, documentation gaps were addressed quickly, and operations returned to normal once the review concluded.
In 2026, this reactive model fails.
CMS contractors increasingly use data analytics to identify patterns that warrant review. This means:
- Audits are targeted, not random
- Review periods may span months or years
- Documentation patterns matter more than isolated claims
Providers that rely on last-minute preparation struggle to respond effectively. Those that maintain continuous audit readiness—through consistent documentation practices, standardized workflows, and accessible records—respond with less disruption and lower risk.
Prior Authorization, Medical Review, and CMS Alignment
Authorization and medical review programs expanded significantly leading into 2026. These programs are designed to control utilization, but they also introduce compliance complexity.
Common failure points include:
- Inconsistent application of authorization rules
- Misalignment between authorization approval and delivered items
- Expired authorizations at time of billing
Operationalizing authorization discipline requires more than tracking approvals. Providers must ensure that authorization parameters align precisely with what is delivered and billed. Automation can support this process, but only when paired with clear ownership and escalation paths.
The Financial Cost of Compliance Failure
Compliance failures carry financial consequences that extend beyond denied claims.
Direct costs include:
- Recoupments from post-payment reviews
- Reduced appeal recovery rates
- Increased staff time spent on corrective action
Indirect costs are often more damaging:
- Cash-flow volatility
- Distorted financial reporting
- Operational disruption during audits
- Erosion of payer trust
Organizations that treat compliance as overhead underestimate its financial impact. In reality, compliance discipline stabilizes revenue by reducing unexpected disruption.
Technology’s Role in CMS Compliance—and Its Limits
Technology plays an important role in compliance, but it is not a substitute for governance.
Automation can:
- Enforce documentation requirements
- Flag missing elements
- Track authorization status
- Support audit trails
However, technology also introduces risk when:
- Workflows are poorly defined
- Exceptions are not reviewed
- Automated decisions lack oversight
CMS holds suppliers accountable regardless of automation. Providers must therefore define clear governance structures that determine where automation applies and where human review is required.
Staff Training, Accountability, and Compliance Culture
Annual compliance training alone is insufficient in today’s environment.
Effective compliance culture requires:
- Role-specific expectations
- Clear accountability for workflow steps
- Reduced reliance on individual judgment
- Ongoing reinforcement of standards
When staff understand not only what is required but why it matters, compliance becomes part of daily work rather than a periodic obligation.
Common CMS Compliance Failure Patterns in 2026
Across the industry, several patterns emerged repeatedly:
- Documentation present but insufficient
- Authorization shortcuts during high volume periods
- Vendor-related compliance gaps
- Inconsistent application of standards across locations
These failures are rarely isolated incidents. They reflect systemic weaknesses that require structural correction.
How Wonder Worth Solutions Helps Providers Stay Compliant
Wonder Worth Solutions supports DME providers by translating CMS requirements into operational controls. By aligning workflows, documentation practices, and monitoring processes with current compliance expectations, organizations reduce risk while maintaining efficiency.
Compliance becomes an asset rather than a liability
Conclusion: Compliance as Infrastructure, Not Overhead
In 2026, CMS compliance is no longer a box to check—it is infrastructure that supports operational stability. DME providers that embed compliance into workflows, reinforce accountability, and maintain continuous readiness position themselves to adapt as rules evolve.
Those that rely on awareness alone will continue to react under pressure.



